WhisperSom™ RestBud™ Device Description and Supporting Research
The prevalence of Obstructive Sleep Apnea (OSA) is estimated to be between 7% and 17% of the adult population in the U.S., or as many as 52,000,000 people.
The current “gold standard” treatment for OSA is Continuous Positive Airway Pressure (CPAP) machines-the technology for which was developed in 1981. CPAP uses a mask and flow generator to apply air pressure to keep the airway open (splint).
An ongoing problem with CPAP is patient utilization. Of those patients for whom CPAP is prescribed it is estimated that some 30% will not use it or quickly give up on it. Another study estimated that of the patients who do use CPAP machines 34% use it intermittently (approximately 4 nights/week), and/or remove it for part of the night (for this group median nightly usage is 3.1 hours).
If those statistics are correct this means that approximately 1 in 2 of all those diagnosed with OSA either do not use CPAP or use it so infrequently that it has no practical efficacy for them.
Our product, WhisperSom RestBud™, has no oral or nasal interfaces, thus avoiding the issues of claustrophobia, entanglement, skin irritation, and air leakage that contribute to the poor compliance levels for CPAP devices. It does not lead to facial deformities (as Mandibular Advancement Devices and CPAP Masks have been accused of doing), and does not involve surgery. It is easy and comfortable to use and adapt to.
Along with the other electronic components RestBud™ contains a suite of sensors that allow the device to perform respiratory auscultation, assess SpO2 saturation levels, and determine the sleeping position of the user.
WhisperSom RestBud™ will directly address what many researchers believe are the causative agents in the development of a number of morbidities/chronic diseases(i.e. Stroke, Heart Attacks, other Cardiac problems, as well as Cancer, Diabetes, Hypertension, Cognitive impairment due to the loss of cerebral grey matter, and non-alcoholic fatty liver disease (NAFLD) – the long term cumulative effects of repetitive occurrences of SpO2 desaturation and large fluctuations of the negative inspiratory intrathoracic pressure.
Most other devices/processes measure their efficacy by a relative improvement in the patient’s Apnea-Hypopnea Index (AHI). It might be more appropriate to measure efficacy by the ability of a device/process to ameliorate the continued accumulation of damage from repetitive Sp02 desaturations. Quantifying how much time is spent with Sp02 desaturations of <95% (some research has shown that this is the level which leads to heart damage (Naresh M. Punjabi et al. (42) and the depth of those desaturations might be a more useful and enlightening metric.
By using that criteria, the only treatment options that can guarantee to maintain Sp02 desaturation of =>95%, and minimize abnormal fluctuations of negative inspiratory intrathoracic pressures during the entire sleeping period, and which is equally efficacious among all levels of severity is CPAP (if used consistently, correctly and appropriately titrated), some surgeries, and the WhisperSom RestBud™.
As of this point in time we have created a Proof of Concept Platform. It has been tested and shown to be efficacious.
Element 1: Detection:
The technology exists to allow the detection of Duty Cycle and Respiration Rate developments that are concurrent with changes to Pharyngeal Patency during the breaths immediately preceding an Obstructive Apnea (55, 56). These changes have been detailed in numerous research papers (1, 2, 3, 4, 8, 35, 44, 57, 59, 62, 73, 82, 89).
This is the results of one study graphically displayed:
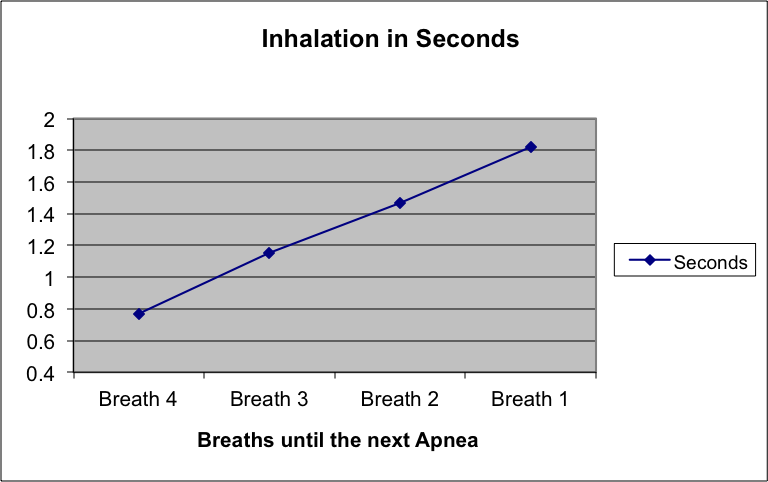
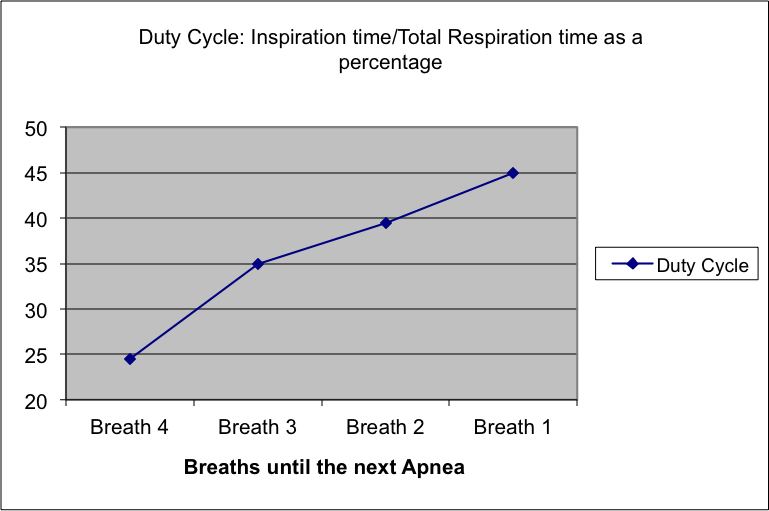
Specifically RestBud™will detect the audio signature of respirations and monitor the Sp02 levels of the user. The data streams from the sensors will allow RestBud™ to determine if the user is facing an actual apnea event, a change to the respiratory duty cycle presaging an apnea event, or a hypopnea that would lead to a Sp02 level <=95%.
The WhisperSom RestBud™ uses heuristic/self adaptive programming, and pattern matching to detect these respiratory changes. The heuristic/self adaptive programming allows RestBud™ to continuously “learn” which patterns an individual user exhibits that precipitate into an obstructive apnea, and adjusts its actions based upon that information.
Element 2: Intervention techniques:
Numerous investigators have applied external stimulation to change ventilation: electrical stimulation applied directly to the hypoglossal nerve, transcutaneously to the submental region, high-frequency oscillation of the upper airway (UA) mucosa to influence the resolution of obstructive apneas (presumably by stimulating UA dilator muscles), tactile stimulus, and most commonly, audio. Specific experiments have determined that audio stimulus is effective at resolving apneas (5, 14) without always causing an EEG/Cortical arousal. In another experiment (69) tone induced arousals were found to have approximately the same duration as spontaneous arousals (approximately 7 seconds) wherein those produced in conjunction with an apnea had a mean length of 12. 6 seconds (70, 74). D. M. Carlson et al. (72) found that acoustically induced cortical arousal increases phasic pharyngeal muscle and diaphragmatic EMG in NREM sleep.
The audio stimulation that most sleep researches use consists of a 500-4KHz tone applied with initial amplitude of 45dB, which is increased to as much105dB in order to trigger the desired effect. It is not uncommon that even at these extreme amplitudes the volunteer is not aroused or awakened. As a reference: a Vacuum Cleaner is rated at 60dB @ six feet while a Motorcycle is rated at 85dB @ twenty-five feet.
Tones are notoriously poor methods for causing a controlled arousal (Children and Smoke Alarms UL Committee, 6). They are uni-dimensional, which while advantageous in a research environment wherein one attempts to minimize variables, are unsuitable for being adapted to subtly applying the required extraneous stimulus to cause an arousal dependent upper airway opening. Indeed research has shown that stimulus reactions are unique to the individual. The problem of deriving individual specific stimulus patterns that rely only on tones are compounded, according to research (79), that shows that there is sleep-specific dampening peculiar to inspiratory effort-related stimuli for those with OSAS.
For those reasons the WhisperSom RestBud™ takes a radically different approach to the use of acoustic stimuli to achieve an arousal-dependant opening, while minimizing stimulus “overshoot” which may lead to EEG/Cortical arousals (which impair sleep quality). It is our belief that the RestBud™ will be superior in provoking non-Cortical arousals than that of the most effective manual methods (80).
- RestBud™ uses novel types of acoustic stimulus. The stimulus is chosen from a library of audio files of different content, amplitude, and length. It is delivered to the user via in-ear monitors.
- By again utilizing heuristic/self adaptive programming RestBud™ will determine which combination of the vast number of controllable parameters afforded by this method of audio stimulus are best suited to meet five goals:
- The prevention of a complete loss of airway patency
- Promoting the restoration of an open airway
- Avoidance of EEG/Cortical arousa5s (14) and awakenings
- Assuring that SpO2 stays =>94%
- Avoiding large fluctuations of the negative inspiratory intrathoracic pressure
The benefit of the heuristic/self adaptive programming is that it will allow for the continuous testing and verification of the effectiveness of the stimulus goals. RestBud™ will always adjust its’ stimulus outflow to match the within night variations in the patient’s arousal index (36, 54).
Some studies have determined that approximately 50% of those diagnosed with mild OSA have positional apneas, i.e. they have are apt to have more apneas while sleeping in the supine position. RestBud™ determines if the user has positional apneas and if so will cause them to shift to a non-supine position.
Element 3: Positive aspects of Intervention:
There exists a number of compelling reasons to avoid an apnea (10, 25). Perhaps the most important is not to disrupt the normalization of blood gases.
The importance of normalization of blood gases was supported in two studies by David W. Hudgel et al. (87, 88) in which it was concluded that changes in the amount of inspired CO2 and O2 had a dramatic impact on the duration of apneas.
During an apnea the SpO2 levels decline (for instance, for a 26 second apnea the decrease was 9.2% (52, 85)), leading to intermittent hypoxia. Hypoxia by itself has been causally linked to many morbidities. However the role of hypoxia in the characteristic increase in blood pressure and heart rate at the end of an apnea episode remains contested with it being supported as a causative agent in some studies (61, 65, 67, 68, 78, 90). A recently released study by Sarah-Jane C Lusina et al. (27) found, “…that 10 daily exposures to isocapnic hypoxia in humans can increase sympathetic outflow during acute hypoxia and recovery”. The significance of increased sympathetic outflow is that it seems to be directly responsible, at least in part, for the acute blood pressure changes that occur at the end of a self-extinguishing apnea event (28, 29, 91). It is the conclusion from a study by Urs A. Leuenberger et al.(30) that, “…these findings strengthen the rationale that preventing intermittent hypoxia, a goal that is presently best achieved with continuous positive airway pressure therapy, is crucial to prevent the adverse neurocirculatory and cardiovascular consequences of sleep apnea.” Another study that links intermittent hypoxia to increased sympathetic outflow was performed by Urs A. Leuenberger et al. (75).
According to Miliauskas et al. (83), “Hypoxemia depth at night is an important factor for daytime sleepiness …”
It is the conclusion of R. Stoohs et al. (52) that hypoxia is a significant mediating agent in changes to Stroke Volume, and Heart Rate. That view is partially supported by a study by C. P. O’Donnell et al. (66).
Conversely a study (64) concluded, “…that only apnea length and pulse rate changes remained significantly related to [blood pressure changes], while hypoxemia did not”. This conclusion also speaks strongly for the case for termination/prevention of an apnea event.
The magnitude of changes to blood pressure varied significantly between degrees of arousal (15, 16, 17, 23, 50, 64, 85) with non-cortical activations (<3s) having a much lower diastolic than that of an EEG/Cortical arousal (>10s). Other studies show that even with acoustically induced EEG/Cortical arousals the magnitude of blood pressure changes is less than those that occur when an apnea self-extinguishes (17, 77). It is a conclusion of O’Driscol et al. (26) that the cardiovascular response occurring at the end of an obstructive apnea is almost double that of which happens after a spontaneous arousal.
The magnitude of the change in heart rate (HR) has also been found to be dependent on the type and magnitude of the arousal. In a study by Amy S. Jordan et al. (50, 51, 85) concluded that the HR change to a spontaneous arousal was +12% over baseline, for an induced arousal from sleep it was +20% over baseline, and for the post apnea tachycardia it was +68% (52). Still another study (70) approximately confirmed those HR changes for spontaneous and induced arousals, while this study (71) detected post apnea changes to HR of more than 80%.
While it has been argued that the repetitive arousals related to apnea events is the proximal cause of the elevated blood pressures (non-dipping) associated with long term OSA a study by Melinda J. Carrington et al. (58) questions that theory as does another study by Dina Brooks et al. (84). If valid, then inducing arousals to terminate incipient apneas should have a minimal effect on dipping.
In a similar vein, hypoxia has been implicated as a risk factor in silent cerebral infarct in a study by Kazuo Eguchi et al.(7).
A study by Naresh M. Punjabi et al. (42) concluded that the frequency of hypopneas associated with a 4.0–4.9% oxyhemoglobin desaturation was associated with prevalent cardiovascular disease after adjusting for the frequency of hypopneas with oxyhemoglobin desaturations above 5%.
It is a conclusion of a study that prior sleep apneas increases the arousal threshold to upper airway occlusion on subsequent nights and prolongs the apnea events (45). Decreasing the quantity and duration of apneas may break that positive feedback cycle (76). Indeed, according to W. De Backer et al. (57) that, “…any intervention that stabilizes the breathing pattern will ultimately also lower the tendency to collapse.”
A study by Danny J. Eckert et al. (60) found that acute sustained hypoxia reduced P1 and P2 amplitudes of the RREP and decreased the sensation of respiratory loading.
The role of hypercapnia is OSA is ambiguous, however in a study by Ayappa et al. (12) “PaCO2 was directly related to apnea/interapnea duration ratio such that with increasing chronic hypercapnia the interapnea duration shortens relative to the apnea duration”.
RestBud™ can help avert intermittent hypoxia and hypercapnia.
Intervention will minimize changes to intrathoracic pressure. Continued respiratory effort after an obstruction leads to increases in negative intrathoracic pressure. Negative intrathoracic pressure encourages venous return to the right ventricle, increases after load on the left ventricle, which causes ventricular hypertrophy and results in congestive heart failure (34).
Studies have shown that there is always a degree of ventilatory overshoot (hyperpnea), be it from a spontaneous, induced arousal, or an apnea related arousal. However the magnitude of the overshoot was found to be significantly larger for an self-extinguishing apnea: In a study by Amy S. Jordan et al. (50, 51) it was determined that the inspiratory minute ventilation change from a spontaneous arousal was +10% over baseline, for an induced arousal from sleep (non apnea) it was +15% over baseline, and from an self-terminating apnea it was 60% (53). If loop gain is the controlling mechanism in the occurrence of repetitive apneas and the amount of PaCO2 is the mediating agent then controlling the degree of hyperpnea should affect the amount of loop gain.
This view is supported in studies that find that hyperventilation is a causal agent for further respiratory disturbances, with the arousal intensity and duration linked to the degree of ventilatory overshoot (35, 46, 86).
In a similar vein some researchers (38, 43, 46) have theorized that the termination of an apnea event by an arousal will invariably lead to periods of repetitive cycling (post hyperventilation apnea). However, other experimental data argues against that conclusion (32, 41, 51, 81, 86).
On the other hand hypoxia is a well-known cause of increased carotid chemoreceptor gain and CNS neurotransmitter turnover which, in turn, appears to lead to apnea promotion (30, 31, 47, 48). Preventing apnea inspired intermittent hypoxia would logically lead to decreased overall loop gain.
Dilation of the airway may be inhibited by the surface tension between the apposing
mucosal surfaces, indeed there exists experimental evidence that an application of a topical lubricant consistently reduced the severity of OSA (39, 40).
RestBud™ will act to restore airway patency before the effect of mucosal surface tension occurs, which if allowed to happen, would require a higher amount of stimulus to overcome.
Element 4: Sleep architecture
While it is generally accepted that any arousal leads to changes in sleep architecture (although the linkage between the AHI and the degree of patient fatigue is not well understood) there exists experimental data that indicates an auditory stimulation which causes a lower level of arousal (CNS) is not as disruptive as that which causes an EEG/Cortical arousal (determined by MSLT and PVT testing) (18).
Stimulation has been found to decrease the frequency of apneas, apnea duration, and changes to sleep architecture with decreased sleep stages I and II, and increased stages III and IV (11). Another study conformed aspects of that study and furthermore found that the improvements reduced the frequency and duration continued for two days. In addition there was an improvement in MSLT scores (13).
In a similar vein another rationale to initiate a controlled arousal through a CNS activation is that the impact upon sleep continuity is
Most patients with OSA develop periods of stable breathing and sleep (36). Younes et al. (38) found that 78% of patients with OSA develop periods of stable breathing and sleep even though there were incidents of complete obstructions (37). Indeed studies have shown that the number of respiratory related arousals (as defined by the AHI) are much lower is Stage 3, Stage 4 (Slow Wave Sleep), and REM (36). Thus most patients currently diagnosed with OSA are capable of dilating their airway without the need of arousal and are receiving restorative sleep.
It was the conclusion of a study by Anne Berssenbrugge et al (62) that, “…attribute(s) the observed pattern of stabilization of breathing with acute restoration of normoxia in hypoxia to the following: the initial transient prolongation of apneic duration results from the increase in the C02-apneic threshold; as PaO2, rises further, the relative ‘gain’ of the peripheral chemoreceptors decreases (reducing the ventilatory overshoot) and PE falls (allowing a rise in PaCO2) both of which act to eliminate apneas and lead to the subsequent stabilization of breathing pattern”.
RestBud™ will promote these periods of natural sleep by ameliorating some of the causes of respiratory instability.
A provocative and confounding issue is the information gathered by studies that seek to measure the impact on sleep by the use of non-invasive ambulatory blood pressure monitoring (APBM). Some conclusions are that, “Sleep is often disturbed by blood pressure monitoring but, irrespective of whether recording provokes arousal, monitored blood pressure is the same.” (19) which was confirmed by (22, 24), another found that, “…86.4% of subjects did not perceive an appreciable sleep deprivation during overnight ABP monitoring despite a frequency of measurements set to 1 every 15 minutes over the entire 24-hour period” (20). The results of that study closely mirrored those reported by Parati G, et al. (21). Both studies relied on questionnaires about the perceived quality of sleep. These studies raise an interesting issue: If ambulatory blood pressure monitoring (with its’ attendant tactile and audio stimulus – cuff inflation/deflation) does lead to arousals then why do those machines not cause changes to blood pressure and perceived sleep deprivation, although a partial explanation is offered in a study (63) that concluded that, “Ambulatory monitoring for 48 consecutive hours reveals a statistically significant pressor response that could reflect a novelty effect in the use of the monitoring device for the first time.” This is all interesting in light of the fact that in many ways the APBM mirrors the stimulus that will be provided by RestBud™.
Element 5: Excessive Daytime Sleepiness (EDS)
While it is our contention that those who refuse conventional treatment options have implicitly decided to live with a degree of excessive daytime sleepiness (EDS), fatigue, and cognitive impairment, and while we are sure that our device resolves the issue of accumulating damage from repetitive SpO2 desaturations, it may not improve sleep architecture (research papers are not definitive). We are pleased to see that there exists some reason to believe that EDS is not as quite as large an issue as many believe (95). It is the conclusion of this study, “…that in this community-based cohort, subjective sleepiness is absent in many individuals…with moderate to severe sleep-disordered breathing.” Vishesh K. Kapur, MD, et al. (93). Another report found that “…a recent review of the Sleep Heart Health Study showed that EDS – considered as a score greater than 10 on the Epworth Sleepiness Scale – was experienced by up to 21% of individuals who did not have sleep apnea, whereas in patients with an apnea hypopnea index (AHI) greater than 30, only 40% of cases experienced sleepiness.” Hernández García (94).
Excerpts from a review by a judge from a SBIR funding application-
Significance: The proposed project is significant in that it provides a simpler therapy for obstructive sleep apnea (OSA) than current treatments such as continuous positive airway pressure (CPAP). If successful, it would be potentially adopted by 12 million patients and commercially profitable.
Approach: The approach may be a good one in that it may be more tolerated than the currently used continuous positive airway pressure (CPAP), which requires the wearing of a pressurized mask and is not acceptable by many patients.
Protection of Human Subjects: There are no significant health risks from use of chest and abdomen belts, or microphone.
Innovation: The project is innovative in that it proposes to replace continuous positive airway pressure (CPAP), the usual treatment for obstructive sleep apnea (OSA), with a simpler system. They propose to detect onset of apnea with sensors, which might include belts around the chest and abdomen to measure ventilation, and a microphone to measure sound. When it detects onset of apnea it would slightly arouse the patient with a vibrator or loudspeaker, thus improving muscle tone in the throat.
Potential Benefits of the Proposed Research to Subjects and Others: If successful, there are potential benefits to subjects.
Importance of Knowledge to be Gained: If successful, there are potential benefits to apnea sufferers.
In Conclusion:
The WhisperSom RestBud™ is potentially a treatment modality that is efficacious, and should be readily accepted by its’ target population.
References
- Respiratory timing during NREM sleep in patients with occlusive sleep apnea.
Ergun Onal and Melvin Lopata
J Appl Physiol. 1986 Oct;61(4):1444-8 - Respiratory mechanics and timing during sleep in occlusive sleep apnea
Richard J. Martin, Bernarde E. Penncock, William C. Orr, Mark H. Sanders, and Roberts M. Rogers
Department of Medicine, University of Oklahoma Health Sciences Center and
Veterans Administration Medical Center, Oklahoma City, Oklahoma 73190
0161-7567/80/0000-0000 Copyright © 1980 the American Physiological Society - Progressive Retropalatal Narrowing Preceding Obstructive Apnea
Mary J. Morrell, Yaseen Arabi, Brian Zahn, And M. Safwan Badr
William S. Middleton Memorial Veterans Hospital, Department of Medicine, and John Rankin Laboratory of Pulmonary Medicine, Department of Preventive Medicine, University of Wisconsin Medical School, Madison, Wisconsin
AM J RESPIR CRIT CARE MED 1998;158:1974–1981. - Inspiratory duty cycle responses to flow limitation predict nocturnal hypoventilation
H. Schneider, V. Krishnan, L. E. Pichard, S. P. Patil, P. L. Smith and A. R. Schwartz
Division of Pulmonary and Critical Care Medicine, Johns Hopkins Sleep Disorders Center, Baltimore, MD, USA.
Eur Respir J 2009; 33:1068-1076 - Effect of induced transient arousal on obstructive apnea duration
C. Basner, E. Onal, D. W. Carley, E. J. Stepanski and M. Lopata
Department of Medicine, Section of Respiratory and Critical Care Medicine, University of Illinois at Chicago College of Medicine, and the Department of Veterans Affairs West Side Medical Center and University of Illinois Hospital, Chicago, Illinois 60612
0161-7567/95 Copyright © 1995 the American Physiological Society - The salience of fire alarm signals for sleeping individuals: A novel approach to signal design
Michelle Ball & Dorothy Bruck
Victoria University, Melbourne, Australia.
In Proceedings of the 3rd International Symposium on Human Behaviour in Fire, Sept 2004, Belfast, Northern Ireland, London: Interscience Communications pp. 303-314.
- Nocturnal Hypoxia Is Associated With Silent Cerebrovascular Disease in High-Risk Japanese Community-Dwelling Population
Kazuo Eguchi, Kazuomi Kario, Satoshi Hoshide, Joji Ishikawa, Masato Morinari, and Kazuyuki Shimmed
Department of Cardiology, Jichi Medical School, Tochigi, Japan
Am J Hypertens (2005) 18, 1489–1495; doi: 10.1016/j.amjhyper.2005.05.032 - Upper Airway Obstruction in Snoring and Upper Airway Resistance Syndrome
Jason P. Kirknessa, Vidya Krishnana, Susheel P. Patila, Hartmut Schneidera
Johns Hopkins University, Department of Medicine, Division of Pulmonary and Critical Care Medicine, Johns Hopkins Sleep Disorders Center, Baltimore, Md., USA; University of Western Australia, School of Anatomy and Human Biology, Faculty of Life and Physical Sciences, Perth, Australia
Prog Respir Res. Basel, Karger, 2006, vol 35, pp 79–89 - Experimentally induced arousals during sleep: a cross-modality matching paradigm
Kato, J. Y. Montplaisir, and G. J . Lavigne
Faculte´s des me´decine and me´decine dentaire, Universite´ de Montre` al, Montre` al, Canada, Centre d_E´ tude sur le Sommeil et Rythmes Biologiques, Hoˆ pital du Sacre`-Coeur de Montre´ al, Montre` al, Canada and Matsumoto Dental University, Institute for Oral Science, Shiojiri, Japan
Sleep Res. (2004) 13, 229–238 - Day-Night Variation of Acute Myocardial Infarction in Obstructive Sleep Apnea
Fatima H. Sert Kuniyoshi, PHD, Arturo Garcia-Touchard, MD, Apoor S. Gami, MD,
Abel Romero-Corral, MD, MSC, Christelle van der Walt, RPSGT, Snigdha Pusalavidyasagar, MD, Tomas Kara, MD, PHD, Sean M. Caples, DO, Gregg S. Pressman, MD, Elisardo C. Vasquez, PHD, Francisco Lopez-Jimenez, MD, MSC, Virend K. Somers, MD, PHD, FACC Rochester, Minnesota; Vitoria, Brazil; Brno, Czech Republic; and Philadelphia, Pennsylvania
Am. Coll. Cardiol. 2008;52;343-346
doi:10.1016/j.jacc.2008.04.027 - A New Treatment for Obstructive Apnea Syndrome by Electrical Stimulation of Submental Region
Hiroshi Miki, Wataru Hida, Hiroshi Inoue And Tamotsut Akishima
The First Department of Internal Medicine, Tohoku
University School of Medicine, Sendai 980
Tohoku J. exp. Med., 1988, 154, 91-92 - Hypercapnia and Ventilatory Periodicity in Obstructive Sleep Apnea Syndrome
Indu Ayappa, Kenneth I. Berger, Robert G. Norman, Beno W. Oppenheimer, David M. Rapoport, and Roberta M. Goldring
Division of Pulmonary and Critical Care Medicine and Bellevue Hospital Chest Service, Department of Medicine, New York University School of Medicine, New York, New York
Am J Respir Crit Care Med Vol 166. pp 1112–1115, 2002
DOI: 10.1164/rccm.200203-212OC - Effects of submental stimulation for several consecutive nights in patients with obstructive sleep apnoea
Wataru Hida, Shinichi Okabe, Hiroshi Miki, Yoshihiro Kikuchi, Osamu Taguchi,
Tamotsu Takishima, Kunio Shirato
First Department of Internal Medicine, Tohoku University School of Medicine,
1-1 Seiryo-machi Aoba-ku, Sendai 980,Japan
Thorax 1994;49:446-452 - Respiratory and Arousal Responses to Acoustic Stimulation
Robert C. Basner, Ergün Önal, Melvin Lopata, Robert Applebaum, and David W. Carley
From the Departments of Medicine and Pharmacology, University of IllinoisCollege of Medicine at Chicago and Veterans Affairs West Side Medical Center, Chicago.
Chest 1997;112;1567-1571 DOI 10.1378/chest.112.6.1567 - Arousal, increased respiratory efforts, blood pressure and obstructive sleep apneoa
Christian Guolleminault and Riccardo Stoohs
Stanford University Sleep Disorders Clinic and Research Center, Stanford, CA
J. Sleep Res. (1995) 4, Suppl. 1, 117-124 - Obstructive sleep apnea syndrome: effect of respiratory events and arousal on pulse wave amplitude measured by photoplethysmography in NREM sleep
José Haba-Rubio, Georges Darbellay, François R. Herrmann, Jean G. Frey, Alda Fernandes, Jean M., Vesin, Jean P. Thiran, and Jean M. Tschopp
Sleep Laboratory, Centre Valaisan de Pneumologie, 3963 Crans-Montana, Switzerland. Institute for Signal Processing Laboratory, Swiss Federal Institute of Technology, Lausanne, Switzerland
Sleep Breath (2005) 9: 73–81
DOI 10.1007/s11325-005-0017-y - Arterial blood pressure responses to graded transient arousal from sleep in normal humans
R. J. 0. Davies, P. J. Belt, S. J. Roberts, N. J. Ali, And J. R. Stradling
Osler Chest Unit, Churchill Hospital, Headington, OxfordOX3 7LJ, United Kingdom
J. Appl. Physiol. 74(3): 1123-1130,1993 - The effect of CNS activation versus EEG arousal during sleep on heart rate response and daytime tests.
C . Guilleminault, V . Abad , P . Philip , R . Stoohs
Clinical Neurophysiology , Volume 117 , Issue 4 , Pages 731 – 739 - Effect on sleep–but not on blood pressure–of nocturnal non-invasive blood pressure monitoring
Schwan A, Eriksson G.
Department of Family Medicine, Uppsala University, Sweden
J Hypertens. 1992 Feb;10(2):189-94; PMID: 1313483 [PubMed] - Ambulatory Blood Pressure and Cardiovascular Outcome in Relation to Perceived Sleep Deprivation
Paolo Verdecchia, Fabio Angeli, Claudia Borgioni, Roberto Gattobigio and Gianpaolo Reboldi
Hypertension 2007;49;777-783; originally published online Jan 29, 2007;
DOI: 10.1161/01.HYP.0000258215.26755.20 - Ambulatory blood pressure monitoring does not interfere with the haemodynamic effects of sleep
Parati G, Pomidossi G, Casadei R, Malaspina D, Colombo A, Ravogli A, Mancia G.
J Hypertens Suppl. 1985 Nov;3(2):S107-9 - Does non-invasive ambulatory blood pressure monitoring disturb sleep
Degaute JP, Van de Borne P, Kerkhofs M, Dramaix M, Linkowski P.
J Hypertens 1992;10:879–85 - Degree of Arousal Is Most Correlated with Blood Pressure Reactivity During Sleep in Obstructive Sleep Apnea
In-Young Yoon,Do-Un Jeong
Yong-In Mental Hospital; Department of Neuropsychiatry and Division of Sleep Studies,
Seoul National University Hospital, Seoul, Korea
J Korean Med Sci 2001; 16: 707-11
ISSN 1011-8934 - Effect of nocturnal blood pressure measurement on sleep and blood pressure during sleep.
Middeke M.
Rhea-Zentrum Spreewald Burg, Germany
Zeitschrift fur Kardiologie 1996;85 Suppl 3():99-105. - Respiratory Disturbances Index:An Independent Predictor of Mortality in Coronary Artery Disease
Yüksel Peker, Jan Hedner, Holger Kraiczi, And Steen Löth
Departments of Pulmonary Medicine and Clinical Pharmacology, Sahlgrenska University Hospital, Gothenburg, Sweden; and Department of Otorhinolaryngology, County Hospital of Skaraborg, Skövde, Sweden
Am J Respir Crit Care Med Vol 162. pp 81–86, 2000 - Cardiovascular response to arousal from sleep under controlled conditions of central and peripheral chemoreceptor stimulation in humans
O’Driscoll, Denise M (DM); Meadows, Guy E (GE); Corfield, Douglas R (DR); Simonds, Anita K (AK); Morrell, Mary J (MJ)
Clinical and Academic Unit of Sleep and Breathing, National Heart and Lung Institute, Imperial College, London SW3 6LY; MacKay Institute of Communication and Neuroscience, School of Life Sciences, Keele University, Keele ST5 5BG;and Sleep and Ventilation Unit, Royal Brompton Hospital, London SW3 6NP, United Kingdom
Appl Physiol 96: 865–870, 2004. First published October 24, 2003; 10.1152/japplphysiol.00749.2003 - Long-term intermittent hypoxia increases sympathetic activity and chemosensitivity during acute hypoxia in humans
Sarah-Jane C Lusina, Paul M Kennedy, J Timothy Inglis, Donald C McKenzie, Najib T Ayas, and A William Sheel
School of Human Kinetics, University of British Columbia, Canada, Faculty of Medicine, University of British Columbia, Canada
J Physiol. 2006 September 15; 575(Pt 3): 961–970. PMCID: PMC1995690 - Effect of episodic hypoxia on sympathetic activity and blood pressure
Fletcher Ec.
Department of Medicine Division of Respiratory, University of Louisville School of Medicine, Ambulatory Care Building, Room A3L01, 530 South Jackson St., Louisville, KY, USA
Respiration Physiology 2000;119(2-3):189-97 - Effects of intermittent hypoxia on sympathetic activity and blood pressure in humans
U.Leuenberger, D.Brubaker, S.Quraishi, C.Hogeman, V.Imadojemu, K.Gray
Division of Cardiology, MC H047, The Pennsylvania State University College of Medicine, The Milton S. Hershey Medical Center, P.O. Box 850, Hershey, PA 17033, United States
Autonomic Neuroscience, 2005, Volume 121, Issue 1, Pages 87-93 - The ventilatory responsiveness to CO2 below eupnoea as a determinant of ventilatory stability in sleep
Jerome A Dempsey, Curtis A Smith, Tadeuez Przybylowski, Bruno Chenuel, Ailiang Xie, Hideaki Nakayama, and James B Skatrud
The John Rankin Laboratory of Pulmonary Medicine, Department of Population Health Sciences, University of Wisconsin-Madison, Madison, WI, 53726-2368, USA
October 1, 2004 The Journal of Physiology, 560, 1-11. - Intermittent hypoxia and respiratory plasticity in humans and other animals: does exposure to intermittent hypoxia promote or mitigate sleep apnoea
Jason H. Mateika, and Gunjan Narwani
Departments of 1Physiology and 3Internal Medicine, Wayne State University School of Medicine, Detroit, MI48201, USA
John D. Dingell Veterans Affairs Medical Center, Detroit, MI48201, USA
Exp Physiol, March 1, 2009; 94(3): 279 – 296. - Influence of arterial O2 on the susceptibility to post-hyperventilation apnea during Sleep
Ailiang Xie, James B. Skatrud, Dominic S. Puleo, and Jerome A. Dempsey
University of Wisconsin, Department of Medicine, Population Health Sciences and the Middleton Memorial Veterans Hospital, Madison, Wisconsin53705, USA
J Appl Physiol (September 22, 2005). doi:10.1152/japplphysiol.00440.2005 - The Relationship Between Esophageal Pressure and Apnea Hypopnea Index in Obstructive Sleep Apnea-Hypopnea Syndrome
Takuya Watanabe, Akira Mikami, Takayuki Kumano-Go, Nakamori Suganuma, Yoshihisa Shigedo, Masamichi Motonishi, Hideharu Honda, Kyoko Kyotani, Shigehiko Uruha, Kiyoji Terashima, Yoshio Teshima, Masatoshi Takeda and Yoshiro Sugita
Department of Medical Science III, School of Health and Sport Sciences, OsakaUniversity, Department of Clinical Neuroscience, Psychiatry, Osaka University Graduate School of Medicine
Sleep Research Online3(4): 169-172, 2000 - Obstructive sleep apnea syndrome and hypertension:mechanism of the linkage and 24-h blood pressure control
Kazuomi Kario
Division of Cardiovascular Medicine, Department of Medicine, Jichi Medical University School of Medicine, Yakushiji, Shimotsuke, Tochigi, Japan
Hypertension Research (2009) 32, 537–541 & 2009 The Japanese Society of Hypertension - Mechanisms used to restore ventilation after partial upper airway collapse during sleep in humans.
Amy S Jordan, Andrew Wellman, Raphael C Heinzer, Yu-Lun Lo, Karen Schorl, Louise Dover, Shiva Gautam, Atul Malhotra and David P White
Sleep Disorders Research Program, Brigham and Women’s Hospital and Harvard Medical School, Boston, USA; General Clinical Research Center, Beth Israel Deaconess
Medical Center, Boston, USA
Thorax 2007;62;861-867; originally published online 5 Apr 2007
doi:10.1136/thx.2006.070300 - Arousability in sleep apnoea/hypopnoea syndrome patients
K. Dingli, I. Fietze, T. Assimakopoulos, S. Quispe-Bravo, C. Witt, N.J. Douglas
Respiratory Medicine Unit, University of Edinburgh, UK; Respiratory Medicine, Charite, Berlin, Germany ;Dynesys Ltd, Edinburgh, UK.
Eur Respir J 2002; 20: 733–740
DOI: 10.1183/09031936.02.00262002 - Role of respiratory control mechanisms in the pathogenesis of obstructive sleep disorders
Magdy Younes
Sleep Centre, Foothills Medical Centre, University of Calgary, Calgary, Alberta, Canada
J Appl Physiol 105: 1389–1405, 2008.
doi:10.1152/japplphysiol.90408.2008. - Contributions of upper airway mechanics and control mechanisms to severity of obstructive apnea
Younes M.
Am J Respir Crit Care Med 168: 645–658, 2003. - Surface Tension Forces in Sleep Apnea: The Role of a Soft Tissue Lubricant
Ruzica Jokic, Artur Klimaszewski, Joseph Mink, And Michael F. Fitzpatrick
Division of Respiratory Medicine, University of Saskatchewan, Saskatoon, Saskatchewan, Canada
Am. J. Respir. Crit. Care Med., Volume 157, Number 5, May 1998, 1522-1525 - Upper airway surface tension: is it a significant cause of airflow obstruction during sleep?
Alan R. Schwartz, Hartmut Schneider, and Philip L. Smith
Johns Hopkins Sleep Disorders Center Johns Hopkins School of Medicine Baltimore, Maryland 21224
J Appl Physiol 95: 1759–1760, 2003; 10.1152/japplphysiol.00747.2003. - Post-hyperventilation hypopnea in humans during NREM sleep
Safwan Badr and Abdulmassih Kawak
Medical Service, William S. Middleton Memorial Veterans Hospital, University of Wisconsin Medical School, Madison, Wisconsin 53705, USA, Department of Medicine, University of Wisconsin Medical School, Madison, Wisconsin 53705, USA, Department of Preventive Medicine, University of Wisconsin Medical School, Madison, Wisconsin 53705, USA
doi:10.1016/0034-5687(95)00083-6
Respiration Physiology Volume 103, Issue 2, February 1996, Pages 137-145 - Sleep-disordered Breathing and Cardiovascular Disease An Outcome-based Definition of Hypopneas
Naresh M. Punjabi, Anne B. Newman, Terry B. Young, Helaine Resnick, and Mark H. Sanders
Division of Pulmonary and Critical Care Medicine, Johns Hopkins University, Baltimore, Maryland; Center for Aging and Population Health, Department of Epidemiology, University of Pittsburgh, Pittsburgh, Pennsylvania; Population Health Sciences, University of Wisconsin; Institute for the Future of Aging Services, American Association of Homes and Services for the Aging, Washington, DC; and Division of Pulmonary and Critical Care Medicine, University of Pittsburgh, Pittsburgh, Pennsylvania
Am J Respir Crit Care Med Vol 177. pp 1150–1155, 2008
Originally Published in Press as DOI: 10.1164/rccm.200712-1884OC on February 14, 2008 - Arousal in the sleep apnea syndrome: a mixed blessing?
W.T. McNicholas
Eur Respir J 1998; 12: 1239–1241
DOI: 10.1183/09031936.98.12061239 - Breath-to-Breath Variability Correlates With Apnea-Hypopnea Index in Obstructive Sleep Apnea
Peter Kowallik, MD; Ilka Jacobi, MD; Alexander Jirmann, MD;Malte Meesmann, MD; Michael Schmidt, MD; and Hubert Wirtz,
CHEST 2001; 119:451–459 - Sleep Apnea Impairs the Arousal Response to Airway Occlusion
Richard B. Berry, Kerry G. Kouchi, Dennis E. Der, Michael J. Dickel and Richard W. Light
Chest 1996;109;1490-1496 DOI 10.1378/chest.109.6.1490 - Ventilatory dynamics during transient arousal from NREM sleep: implications for respiratory control stability
Michael C. K. Keioo, Steve S. W. Koh, John J. W. Shin, Philip R. Westbrook, And Richard B. Berry
Biomedical Engineering Department, University of Southern California, Los Angeles 90089-1451; Department of Medicine, University of California, Irvine 92664; Veterans Affairs Medical Centez; Long Beach 90822; and Pacific Sleep Medicine, Redlands, California 92373
Appl. Physiol. 80(5): 14751484,1996. - Apnea and Periodic Breathing during Sleep
Neil S. Cherniack, M.D.
University of Medicine and Dentistry of New Jersey, Newark, NJ07103-2714
The New England Journal of Medicine — September 23, 1999 — Vol. 341, No. 13 (Editorial) - Posthypoxic ventilatory decline during NREM sleep: influence of sleep apnea
Amal M. Omran, Salah E. Aboubakr, Loutfi S. Aboussouan, Lisa Pierchala, and M. Safwan Badr
Medical Service, John D. Dingell Veterans Affairs Medical Center, and Division of Pulmonary Critical Care Medicine,
Department of Medicine, Wayne State University School of Medicine, Detroit, Michigan48201
J Appl Physiol 96: 2220–2225, 2004. - Role of Arousals in the Pathogenesis of Obstructive Sleep Apnea
Magdy Younes
Department of Medicine, University of Manitoba, Winnipeg, Manitoba; and Department of Medicine, University of Toronto, Toronto, Ontario, Canada
Am J Respir Crit Care Med Vol 169. pp 623–633, 2004
DOI: 10.1164/rccm.200307-1023OC on December 18, 2003 - Ventilatory Response to Brief Arousal from Non–Rapid Eye Movement Sleep Is Greater in Men Than in Women
Amy S. Jordan, Danny J. Eckert, Peter G. Catcheside and R. Doug McEvoy
Adelaide Institute for Sleep Health, Repatriation General Hospital, Daw Park; Department of Physiology, University of Adelaide, Adelaide; and School of Medicine, Flinders University, Bedford Park, South Australia, Australia
Am J Respir Crit Care Med Vol 168. pp 1512–1519, 2003
Originally Published in Press as DOI: 10.1164/rccm.200302-150OC on October 2, 2003 - The influence of gender and upper airway resistance on the ventilatory response to arousal in obstructive sleep apnoea in humans.
Jordan AS, McEvoy RD, Edwards JK, Schory K, Yang CK, Catcheside PG, Fogel RB, Malhotra A, White DP.
Brigham and Women’s Hospital, Sleep Disorders Research Program @ BIDMC, 75 Francis Street, Boston, MA02115
J Physiol. 2004 Aug 1;558(Pt 3):993-1004. Epub 2004 Jun 24 - Cardiovascular changes associated with obstructive sleep apnea syndrome
Stoohs and C. Guilleminault
Sleep Disorders Clinic and Research Center, Stanford University School of Medicine, California94305.
J Appl Physiol 72: 583-589, 1992 - Selective Potentiation of Peripheral Chemoreflex Sensitivity in Obstructive Sleep Apnea
Krzysztof Narkiewicz, Philippe J. H. van de Borne, Catherine A. Pesek, Mark E., Dyken, Nicola Montano and Virend K. Somers
From the Cardiovascular Division, Department of Internal Medicine (K.N., P.J.H.v.d.B., C.A.P., V.K.S.) and Department of Neurology (M.E.D.),University of Iowa College of Medicine, Iowa City, and Centro L.I.T.A. Vialba, Centro Ricerche Cardiovascolari, CNR, Medicina Interna II, Ospedale` L. Sacco, Universita´ degli Studi di Milano, Italy (N.M.).
Circulation 1999;99;1183-1189 - Within-night variation in respiratory effort preceding apnea termination and EEG delta power in sleep apnea
Richard B. Berry, Musa A. Asyali, Michael I. McNellis, and Michael C. K. Khoo
Department of Medicine, Veterans Affairs Medical Center, Long Beach 90822; University of California, Irvine 92717; and Department of Biomedical Engineering, University of Southern California, Los Angeles, California 90033
J. Appl. Physiol. 85(4): 1434– 1441, 1998 - Fuzzy assessment of sleep-disordered breathing during continuous positive airway pressure therapy.
Shin JJ, Berry RB, Khoo MC.
Biomedical Engineering Department, University of Southern California, Los Angeles90089-1451, USA.
Sleep. 1998 Dec 15;21(8):817-28. - Pattern recognition of obstructive sleep apnoea and Cheyne–Stokes respiration
Gerhard Weinreich, Jeff Armitstead, and Helmut Teschler
Ruhrlandklinik, Department of Pneumology, University Hospital, Tuschener Weg 40, D-45239 Essen, Germany; Graduate School of Biomedical Engineering, University of New South Wales, Sydney 2052, Australia
Physiol. Meas. 29 (2008) 869–878 - Obstructive Sleep Apnea-Hypopnea Syndrome Definitions and Pathophysiology
Randerath WJ, Sanner BM, Somers VK
Department of Pulmonary Medicine, University of Antwerp, Edegem, Belgium
Prog Respir Res. Basel, Karger, 2006, vol 35, pp 90–96 - Blood Pressure and Heart Rate During Continuous Experimental Sleep Fragmentation in Healthy Adults
Melinda J. Carrington, PhD; John Trinder, PhD
Department of Psychology, The University of Melbourne, Victoria, Australia; Preventative Cardiology, Baker Heart Research Institute, Victoria, Australia
SLEEP, Vol. 31, No. 12, 2008 - The effects of hypoxia on load compensation during sustained incremental resistive loading in patients with obstructive sleep apnea
Michael C. Hlavac, Peter G. Catcheside, Amanda Adams, Danny J. Eckert, andR. Doug McEvoy
Adelaide Institute for Sleep Health, Repatriation General Hospital, Daw Park, South Australia; Department of Medicine, Flinders University, Bedford Park, South Australia; and School of Molecular and Biomedical Science, Discipline of Physiology, University of Adelaide, South Australia, Australia
J Appl Physiol 103: 234–239, 2007.
First published April 5, 2007; doi:10.1152/japplphysiol.01618.2005. - Sustained Hypoxia Depresses Sensory Processing of Respiratory Resistive Loads
Danny J. Eckert, Peter G. Catcheside, Rachel McDonald, Amanda M. Adams, Kate E. Webster, Michael C. Hlavac, and R. Doug McEvoy
Adelaide Institute for Sleep Health, Repatriation General Hospital, Daw Park, South Australia; School of Molecular and Biomedical Science, Discipline of Physiology, University of Adelaide, South Australia; Department of Medicine, Flinders University, Bedford Park, South Australia; and Musculoskeletal Research Centre, La Trobe University, Bundoora, Victoria, Australia
Am J Respir Crit Care Med Vol 172. pp 1047–1054, 2005
Originally Published in Press as DOI: 10.1164/rccm.200505-699OC on June 23, 2005 - Daytime blood pressure elevation after nocturnal hypoxia
Yaseen Arabi, Barbara J. Morgan,Brian Goodman, Dominic S. Puleo, Ailiang Xie, and James B. Skatrud
Departments of Medicine and Surgery, University of Wisconsin, Madison 53792; and William S. Middleton Memorial Veterans Hospital, Madison, Wisconsin53705
J Appl Physiol 87:689-698, 1999 - Mechanisms Of Hypoxia-Induced Periodic Breathing During Sleep In Humans
Anne Berssenbrugge, Jerome Dempsey, Conrad Ibert, James Skatrudt and Pamela Wilson
The John Rankin Laboratory of Pulmonary Medicine, Department of Preventive Medicine, University of Wisconsin, Madison, WI 53705, U.S.A. and the William S. Middleton Memorial Veterans Administration Hospital, Madison, WI 53705, U.S.A.
J Physiol. (1983), 343, pp. 507-524 - Evaluation of the Extent and Duration of the “ABPM Effect” in Hypertensive Patients
Ramo´n C. Hermida, PhD, Carlos Calvo, MD, PhD, Diana E. Ayala, MD, PhD, Jose R. Fernandez, PhD, Luis M. Ruilope, MD, PH\hD, Jose´ E. Lopez, MD
Vigo, Santiago de Compostela, and Madrid, Spain
Journal of the American College of Cardiology Vol. 40, No. 4, 2002 © 2002 by the American College of Cardiology Foundation - The acute effects of continuous positive airway pressure and oxygen administration on blood pressure during obstructive sleep apnea.
N J Ali, R J Davies, J A Fleetham, and J R Stradling
Chest 1992;101;1526-1532 DOI 10.1378/chest.101.6.1526 - Decrease in ventricular stroke volume at apnea termination is independent of oxygen desaturation
E. Garpestad, J. A. Parker, H. Katayama, J. Lilly, T. Yasuda, J. Ringler, H. W. Strauss and J. W. Weiss
Charles A. Dana Institute, Beth Israel Hospital, Boston, Massachusetts.
J Appl Physiol 77: 1602-1608, 1994 - Airway obstruction during sleep increases blood pressure without arousal
C. P. O’Donnell, T. Ayuse, E. D. King, A. R. Schwartz, P. L. Smith, and J. L. Robotham
Pulmonary Anesthesia Laboratory, Department of Anesthesiology and Critical Care Medicine, and Division of Pulmonary and Critical Care Medicine, Department of Medicine, Johns Hopkins University, Baltimore, Maryland, 21224
J. Appl. Physiol. 80(3): 773-781, 1996.-Recent - Surges of muscle sympathetic nerve activity during obstructive apnea are linked to hypoxemia.
Leuenberger U, Jacob E, Sweer L, Waravdekar N, Zwillich C, Sinoway L.
Department of Medicine, Milton S. Hershey Medical Center, Pennsylvania State University, Hershey, PA17033, USA
J Appl Physiol 1995;79(2):581-8 - Role of hypoxemia in sleep apnea-induced sympathoexcitation.
Smith Ml, Niedermaier ON, Hardy SM, Decker MJ, Strohl KP.
Department of Medicine, Case Western Reserve University, Cleveland, OH44106, USA
Journal of the Autonomic Nervous System 1996; 56(3):184-90. - Cardiac changes during arousals from non-REM sleep in healthy volunteers
Eugene Nalivaiko, Peter G. Catcheside, Amanda Adams, Amy S. Jordan, Danny J. Eckert, and R. Doug McEvoy
Department of Human Physiology and Centre for Neuroscience; Department of Medicine, Flinders University; and Adelaide Institute for Sleep Health, Repatriation General Hospital, Adelaide, Australia
Am J Physiol Regul Integr Comp Physiol 292: R1320–R1327, 2007 - Variation in the Arousal Pattern after Obstructive Events in Obstructive Sleep Apnea
John R. Stradling, Debby J. Pitson, Lesley Bennett, Charles Barbour, and Robert J. O. Davies
Osler Chest Unit, Churchill Hospital, Oxford, United Kingdom
Am. J. Respir. Crit. Care Med., Volume 159, Number 1, January 1999, 130-136 - Age Related Differences in Electroencephalographic and Cardiac Arousal at the Termination of Sleep Apnea/Hypopnea
Akiko Noda, Fumihiko Yasuma, Tamotsu Okada, Yasuo Koike, Nobuo Nakashima, and Mitsuhiro Yokota
From Nagoya University School of Health Sciences, the First Department of Internal Medicine, the Department of Clinical Laboratory Medicine, Nagoya University Hospital and Okada Clinic, Nagoya
Internal Medicine Vol. 39, No. 5 (May 2000) - Acoustically induced cortical arousal increases phasic pharyngeal muscle and diaphragmatic EMG in NREM sleep
D. M. Carlson, D. W. Carley, E. Onal, M. Lopata and R. C. Basner
Department of Medicine, University of Illinois College of Medicine at Chicago.
Journal of Applied Physiology, Vol 76, Issue 4 1553-1559 - Detection of inspiratory flow limitation during sleep by computer assisted respiratory inductive plethysmography
V. Kaplan, J.N. Zhang, E.W. Russi, K.E. Bloch
Pulmonary Division, Dept of Internal Medicine, University Hospital of Zuerich, Zuerich, Switzerland.
Eur Respir J 2000; 15: 570±578 - Variation in the duration of arousal in obstructive sleep apnea.
Nigro, Carlos Alberto (CA); Rhodius, Edgardo Edmundo
Sleep Laboratory, Hospital Alemán, Buenos Aires, Argentina
Med Sci Monit 2005-Apr; vol 11 (issue 4) : pp CR188-92 - Short-term intermittent hypoxia enhances sympathetic responses to continuous hypoxia in humans
Urs A. Leuenberger, Cynthia S. Hogeman, Sadeq Quraishi, Latoya Linton-Frazier, and Kristen S. Gray
Penn State Heart and Vascular Institute, The Pennsylvania State University College of Medicine, The Milton S.Hershey Medical Center, Hershey, Pennsylvania
J Appl Physiol 103: 835–842, 2007. - Sleep Apnea Impairs the Arousal Response to Airway Occlusion
Richard B. Berry, MD, FCCP; Kerry G Kouchi, AA; Dennis E. Der, BS; Michael J. Dickel, PhD; and Richard W. Light, MD, FCCP
From the Department of Medicine, Long Beach VA Medical Center, University of California Irvine.
Chest 1996;109;1490-1496 - Arousals and aircraft noise – environmental disorders of sleep and health in terms of sleep medicine
F Raschke
Institute of Rehabilitation Research at Norderney, Clinic of the University of Witten/Herdecke, Norderney, Germany
Noise Health 2004;6(22):15-26. - Repetitive apneas induce periodic hypertension in normal subjects through hypoxia
Joost G. Van Den Aardweg and John M. Karemaker
Departments of Internal Medicine and Physiology, Academisch Medisch Centrum,
1105 AZ Amsterdam, The Netherlands
J Appl. Physiol. 72(3): 821-827,1992. - Sleep and respiratory stimulus specific dampening of cortical responsiveness in OSAS.
Afifi L, Guilleminault C, Colrain IM.
Department of Neurology, Cairo University, Cairo, Egypt.
Respir Physiol Neurobiol. 2003 Jul 16;136(2-3):221-34 - The effect of nonvisible sleep fragmentation on daytime function
SE Martin, PK Wraith, IJ Deary and NJ Douglas
Department of Medicine, University of Edinburgh, United Kingdom.
Am. J. Respir. Crit. Care Med., Vol 155, No. 5, May 1997, 1596-1601. - Periodicity of Obstructive Sleep Apneain Patients With and Without Heart Failure
Clodagh M. Ryan, MB; and T. Douglas Bradley, MD
Chest 2005;127;536-542
DOI 10.1378/chest.127.2.536 - Effect of Resistive Loading on Variational Activity of Breathing
Thomas Brack, Amal Jubran, and Martin J. Tobin
Division of Pulmonary and Critical Care Medicine, Edward Hines Jr. Veterans Administration Hospital; and Division of Pulmonary and Critical Care Medicine, Loyola University of Chicago Stritch School of Medicine, Hines, Illinois
Am J Respir Crit Care Med Vol 157. pp 1756–1763, 1998 - Peculiarities of nocturnal oxygen saturation in obstructive sleep apnea
Miliauskas S, Sakalauskas R.
Department of Pulmonology and Immunology, KaunasUniversity of Medicine,
Eiveniu 2, 50009 Kaunas, Lithuania
Medicina (Kaunas) 2005;41(3):217-20. - Obstructive Sleep Apnea as a Cause of Systemic Hypertension. Evidence from a Canine Model
Dina Brooks, Richard L. Horner, Louise F. Kozar, Caroline L. Render-Teixeira, and Eliot A. Phillipso
Department of Medicine, University of Toronto, Toronto, Ontario, Canada, M5S 1A8,
Clin. Invest. 1997. 99:106–109. - Obstructive sleep apnea syndrome: effect of respiratory events and arousal on pulse wave amplitude measured by photoplethysmography in NREM sleep
José Haba-Rubio, Georges Darbellay, François R. Herrmann, Jean G. Frey, Alda Fernandes, Jean M. Vesin, Jean P. Thiran, Jean M. Tschopp
Sleep Breath (2005) 9: 73–81 DOI 10.1007/s11325-005-0017-y - Ventilatory response to induced auditory arousals during NREM sleep.
Badr Ms, Morgan BJ, Finn L, Toiber FS, Crabtree DC, Puleo DS, Skatrud JB.
William S. Middleton Memorial Veterans Hospital, Department of Medicine, University of Wisconsin Medical School, Madison, USA
Sleep 1997;20(9):707-14 - Alteration in obstructive apnea pattern induced by changes in O2 and CO2 inspired concentrations
Hudgel DW, Hendricks C, Dadley A.
Department of Medicine, Case Western Reserve University, Cleveland, Ohio.
Am Rev Respir Dis. 1988 Jul;138(1):16-9. - Instability of Ventilatory Control in Patients with Obstructive Sleep Apnea
David W. Hudgel, Elizabeth A. Gordon, Sitthep Thanakitcharu, and Eugene N. Bruce
Case Western Reserve University Metro Health Medical Center, Cleveland, Ohio; and Center for Biomedical Engineering, University of Kentucky, Lexington, Kentucky
Am. J. Respir. Crit. Care Med., Volume 158, Number 4, October 1998, 1142-1149 - The changes of respiratory mechanics in patients with obstructive sleep apnea-hypopnea syndrome during sleep
Ye H, Guo Xh
Institute of Respiratory Medicine; Beijing Chaoyang Hospital affiliated to the Capital University Of Medical Sciences, Beijing 100020, China
Zhonghua Jei He He Hu Xi Za Zhi 2008; 31 (1):26-31 - Sympathetic Activity and Blood Pressure in the Sleep Apnea Syndrome
Eugene C. Fletcher
Department of Medicine, Division of Respiratory, Critical Care, and Environmental Medicine, Louisville Veterans Affairs Medical Center, and Department of Medicine, University of Louisville School of Medicine, Louisville, Kentucky
Published in Respiration Vol. 64(Suppl 1), pp 22-28, 1997 - Neurocirculatory consequences of negative intrathoracic pressure vs. asphyxia during voluntary apnea
J. Morgan, T. Denahan and T. J. Ebert
Department of Kinesiology, University of Wisconsin, Madison 53706.
Journal of Applied Physiology, Vol 74, Issue 6 2969-2975, Copyright © 1993 by American Physiological Society - On the Potential Clinical Relevance of the Length of Arousals From Sleep in Patients With Obstructive Sleep Apnea
Daniel J. Schwartz, M.D.; Pat Moxley, R.P.S.G.T
The Tampa Sleep Center, University Community Hospital, and Department of Medicine, University of South Florida, Tampa, FL
Journal of Clinical Sleep Medicine, Vol. 2, No. 2, 2006 - Sleepiness in Patients with Moderate to Severe Sleep-Disordered Breathing
Vishesh K. Kapur, MD; Carol M. Baldwin, RN, PhD; Helaine E. Resnick, PhD; Daniel J. Gottlieb, MD; F. Javier Nieto, MD, PhD
Department of Medicine, University of Washington, Seattle, WA; Arizona State University College of Nursing (Southwest Borderlands), Tempe, AZ; MedStar Research Institute, Hyattsville, MD; Department of Medicine, Boston University and the VA Boston HCS, Boston, MA; Department of Population Health Science, University of Wisconsin, Madison, WI
SLEEP, Vol. 28, No. 4, 2005 - Sleep Apnea-Hypopnea Syndrome Without Excessive Daytime Sleepiness
Hernández García
Servicio de Neumología, Hospital Universitario de Canarias, Santa Cruz de Tenerife, Spain
2008 SEPAR - Excessive Daytime Sleepiness in Sleep Apnea: It’s Not Just Apnea Hypopnea Index
Alexandros N. Vgontzas, MD
Sleep Research and Treatment Center, Department of Psychiatry H073, Penn State College of Medicine,500 University Drive, Hershey, PA 17033
Sleep Med. 2008 October; 9(7): 712–714. doi:10.1016/j.sleep.2008.05.001.
